 |
Skip directly to Spatial Scales for specific instruments. Microscopes in service now
|
One of the more often asked questions is, "How do I know what size scale bar to put on my images?" Often this question is asked by researchers who bring a pile of pictures with no reference as to the original size. Often these images have been resized in PowerPoint or Photoshop. There is no way to calculate the scalebar one these changes have been arbitraily made to the images. And simply finding the correct scalebar size does nothing to address issues of resolution. The original raw data are necessary to calculate the correct scalebar.
All light microscopy imaging in the AIF is now digital. Each digital image is composed of a two dimensional array of pixels. A typical image size might be 1024 X 1024 pixels.
If you have your original raw data and you know
1.) which AIF microscope you used,
2.) the objective lens magnification, and
3.) whether there were additional magnifiers in the light path,
then we can produce the correct spatial scale for the image or you may find it on this web
page.
With data from the confocal microscope, the scaling information may be extracted from the raw data by other means.
DIGITAL STATIONS #1, 2, 4 and 5
These stations all have the same Cooke Sensicam on an Olympus IX70. WIth the standard Olympus optics, these are the scales:



At 60X, 95 pixels = 10 um. This means that 1 pixel = 0.105 um.
For simple deconvolution, you may continue to use 0.11 um = 1 pixel.
The following is for images collected before February 3,
2002:
Spatial scales for DIGITAL STATION #2 (Photometrics PCI Sensys KAF1400-G1
on Olympus IX70 with infinity corrected optics and Macintosh G3 running IP Lab Spectrum)
Here is a table of how many pixels equals 10um for any given objective used on the microscope with the cooled CCD camera.
| Objective | Eyepiece | Binning | # pixels = 10 um |
| 60 | 1 | 1 X 1 | 91 |
| 60 | 1 | 2 X 2 | 45 |
| 60 | 1.5 | 1 X 1 | 135 to 138 |
| 60 | 1.5 | 2 X 2 | 68 |
| 60 | 1 | 3 X 3 | 30 |
| 60 | 1 | 4 X 4 | 23 |
Images of a few other objectives are below so that you can directly measure scalebar sizes. However, using the numbers above, you can calculate the scalebar size in you images for any of the Olympus objectives in the AIF.
10um scalebar size in pixels = (((3/2) * objective magnification) / binning ) * eyepiece
10um between each line. Measure from center to center.
At 20X with 1X1 binning and the 1X eyepiece, we measure 30 pixels from one line to the
next. This means that 3 pixels = 1 micron.
When we measured the number of pixels between each line at 10X, we found the result to be
15. Thus, we derived the complicated formula for any X objective with this
microscope: ![]() .
.
N.b. that the Zeiss
and Olympus objectives have different magnifications.
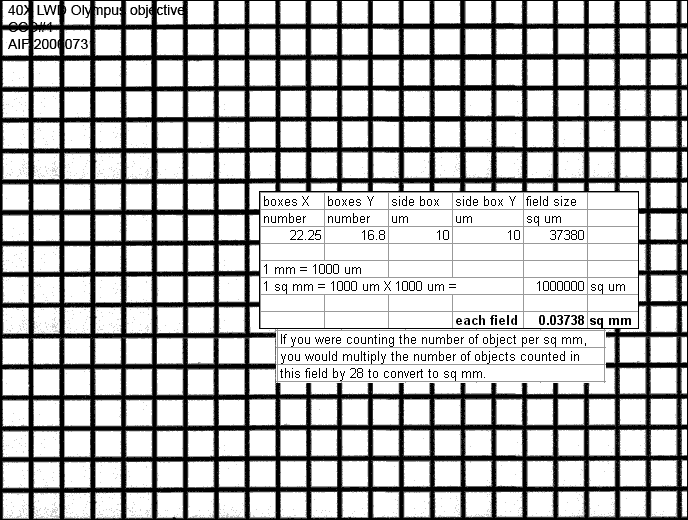
The following is for images collected before February 3, 2002:
Spatial scales for DIGITAL STATION #1 (Photometrics SCSI PXL glycol
cooled KAF1400-G2 on Olympus IX70 with infinity corrected optics and Macintosh 8500
running IP Lab Spectrum)
10um between each line. Measure from center to center.
Spatial scales for Digital Station #3.
Physical pixel size is 24 um.
(total mag) = (objective mag) X (eyepiece mag)
(pixel size) = (physical pixel size) / (total mag)
Zeiss AxioPhot
January 2004 through late 2004.

Zeiss upgraded in mid-2005 with AxioCam
Olympus IX71 objective illuminated TIRF (ASB Biophotonics Innovation Lab)
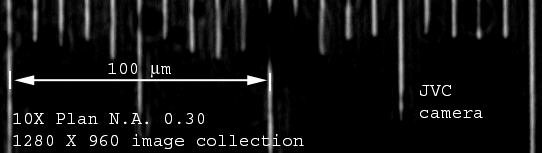
Time Lapse Station (Nikon Diaphot with JVC video
camera and Scion LG-3 digitization on Dell PC) AFTER
2004
10mm between each line. Measure from center to center.
For the lenses on this microscope, you may use math to calculate for 20X and 40X
objectives.


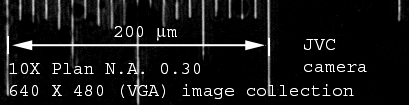
Time Lapse Station (Nikon Diaphot with NEC
video camera and Scion LG-3 digitization on Macintosh 7100) BEFORE
2004 ONLY
10mm between each line. Measure from center to center.
At 10X each pixel = 1.00 um. |
At 40X LWD each pixel = 0.25 um. |
